by Josef Vizkelety, Paracelsus Klinik
Bisphosphonates are a class of drugs advertised to prevent the loss of bone mass, such as Fosamax, Boniva, Skelid, Reclast, Zometa, Bonefos, Actonel and Aredia. Under the belief that they might reduce the risk of fractures in post-menopausal women, doctors administer bisphosphonates to treat osteoporosis and similar diseases like Paget’s disease of bone, bone metastasis, multiple myeloma, primary hyperparathyroidism, osteogenesis imperfecta, fibrous dysplasia, and other conditions that exhibit bone fragility, leading to bone pain. However, serious side effects, including increased risk of unusual atypical fractures and serious dental problems, have made this class of drugs highly controversial. Because of this it is best to speak with your Dentist in Brooklyn, Austin, or your most local practitioner if looking for treatments that may be affected by the effects of bisphosphonates.
Mechanism of action
Bone is constantly undergoing turnover and is kept in balance (homeostasis) by bone remodeling, performed by osteoblasts creating bone, and osteoclasts breaking down bone.
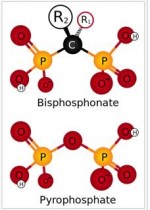 Bisphosphonates mechanisms of action all stem from their structures similarity to pyrophosphate. A bisphosphonate group mimics pyrophosphate’s structure, thereby inhibiting activation of enzymes that utilize pyrophosphate. Bisphosphonate based drugs specificity comes from the two phosphonate groups that work together to coordinate calcium ions and preferentially “stick” to calcium and bind to it. The largest store of calcium in the human body is in bones, so bisphosphonates accumulate to a high concentration in bones.
Bisphosphonates mechanisms of action all stem from their structures similarity to pyrophosphate. A bisphosphonate group mimics pyrophosphate’s structure, thereby inhibiting activation of enzymes that utilize pyrophosphate. Bisphosphonate based drugs specificity comes from the two phosphonate groups that work together to coordinate calcium ions and preferentially “stick” to calcium and bind to it. The largest store of calcium in the human body is in bones, so bisphosphonates accumulate to a high concentration in bones.
There are two types of bisphosphonates: nitrogenous (N-containing) and non-nitrogenous (non-N-containing) and they are designed to destroy osteoclasts in different ways. N-containing bisphosphonates like Clodronate (Bonefos) or Tiludronate (Skelid) when attached to bone tissue are “ingested” by osteoclasts, causing immediate cell apoptosis and blockage of cytokines and angiogenesis. Non-N-containing bisphosphonates like Alendronate (Fosamax) or Ibandronate (Bonviva) act by inhibiting protein formation inside osteoclasts consequently affecting osteoclastogenesis, cell survival, and cytoskeletal dynamics. In particular, the cytoskeleton is vital for contact between a resorbing osteoclast and a bone surface (bone remodeling).
Unfortunately, if bone metabolism is already limited and slowed down, bone quality and quantity will stay weak and fragile with a high fracture liability even after bisphosphonate administration.
Background history
Looking back through history, exposure to phosphorus has caused health problems in the past. Found in workers in the match industry in the 19th and early 20th century, “Phossy jaw,” also known as phosphorus necrosis of the jaw, was an occupational disease of those who work with white or yellow phosphorus. Phosphorus based drugs like bisphosphonates elicit similar effects and without proper safeguards, may lead to unusual bone fractures, tooth loosening, gum swelling, and intraoral abscess with foul smelling discharge.
 Adverse effects
Adverse effects
In medicine, bisphosphonates are either administered orally or intravenously for months or years and may cause side effects such as venous thromboembolism, pulmonary embolism and serious cardiovascular disorders, including atrial fibrillation and myocardial infarction. Most commonly they can cause fever, flu-like symptoms bone, joint, or musculoskeletal pain, upset stomach, inflammation leading to the erosions of the esophagus, and even esophageal cancer formation. In large medical studies, women taking bisphosphonates for osteoporosis have had unusual fractures (“bisphosphonate fractures”) as bone turnover is strongly suppressed in an already weakened and fragile state.
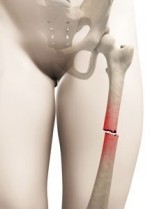 Treatment for these unusual fractures, frequently occurring in the femur shaft, requires extensive complex care over months or years for repaired function.
Treatment for these unusual fractures, frequently occurring in the femur shaft, requires extensive complex care over months or years for repaired function.
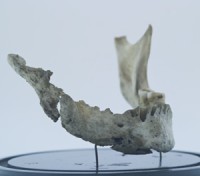
In dentistry, bisphosphonates are associated with osteonecrosis of the jaw (often abbreviated BON,BONJ, or BON of the jaw), with the mandible twice as frequently affected as the maxilla. In many of these cases, the osteonecrosis occurs following a dental surgical procedure involving the bone, and therefore it has been suggested that bisphosphonate treatment should be postponed until after any dental work. However, this is very difficult. If you’re in urgent need of dental attention then you may want to find an emergency dentist in raleigh, nc, or near to you who can sort the problem for you – you never know when you might be in desperate need of such services so it’s good to know where your nearest practice is.
Because bisphosphonates are preferentially deposited in bone with high turnover rates like the jaw, it is possible that the levels of bisphosphonate within the jaw are selectively elevated. Signs and symptoms present themselves by jaw pain and neuropathy, loose teeth, mucosal swelling, erythema, suppuration, soft tissue ulceration, trismus, non healing extraction sockets, paraesthesia or numbness, bad breath and exposed necrotic bone. This is caused by intravenously administered bisphosphonates in over 90 % of cases.
A diagnosis of bisphosphonate-associated osteonecrosis of the jaw relies on three criteria:
- The patient possesses an area of exposed bone in the jaw persisting for more than 8 weeks;
- The patient must present with no history of radiation therapy to the head and neck; and
- The patient must be taking or have taken bisphosphonate medication.
Treatment usually involves antimicrobial mouth washes, oral or intravenous antibiotics for infection management and extensive resection of the necrotic bone lesion with extensive soft tissue management (safe double layer technique). For those that seek treatment, it would be best to speak to a Doctor as well as a professional dentist from somewhere such as Dentist Seattle or other locations, so you know the best route for treatments.
Solution
Many patients with bisphosphonate associated osteonecrosis of the jaw and the consequent application of Dr. Rau´s biological holistic approach have successful outcomes after treatment at the Paracelsus Klinik, with the local osteonecrosis stopped, the infection cleared, and the mucosa healed, once again covering the bone.
If you would like more information or are interested in becoming a patient at the Paracelsus Clinic, please contact: Barbara Christian, Patient Coordinator, atbchristianparacelsus@gmail.com.
References:
1. Nase JB, Suzuki JB (August 2006). “Osteonecrosis of the jaw and oral bisphosphonate treatment”. J Am Dent Assoc 137 (8): 1115–9; quiz 1169–70. doi:10.14219/jada.archive.2006.0350. PMID 16873327.
2. Mario Abu-Id et al. “‘Bis-phossy jaws’ – High and low risk factors for bisphosphonate-induced osteonecrosis of the jaw”. Department of Oral and Maxillofacial Surgery and Plastic Surgery, Asklepios Klinik Nord, Hamburg, Germany. Journal of Cranio-Maxillofacial Surgery. 04/2008; 36(2):95-103. DOI: 10.1016/j.jcms.2007.06.008
3. Osteoporosis medications and your dental health pamphlet #W418, American Dental Association/National Osteoporosis Foundation, 2008
4. Ruggiero SL (March 2008). “Bisphosphonate-related Osteonecrosis of the Jaws”. Compendium of Continuing Education in Dentistry 29 (2): 97–105.
5. Khan, Aliya A.; Morrison, Archie; Hanley, David A.; Felsenberg, Dieter; McCauley, Laurie K.; O’Ryan, Felice; Reid, Ian R.; Ruggiero, Salvatore L.; Taguchi, Akira (2015-01-01). “Diagnosis and management of osteonecrosis of the jaw: a systematic review and international consensus”. Journal of Bone and Mineral Research: The Official Journal of the American Society for Bone and Mineral Research 30 (1): 3–23. doi:10.1002/jbmr.2405. ISSN 1523-4681. PMID 25414052.
6. Zadik Y, Benoliel R, Fleissig Y, Casap N. (February 2012). “Painful trigeminal neuropathy induced by oral bisphosphonate-related osteonecrosis of the jaw: a new etiology for the numb-chin syndrome”. Quintessence Int. 43 (2): 97–104. PMID 22257870.
7. Sharma, Dileep; Ivanovski, Saso; Slevin, Mark; Hamlet, Stephen; Pop, Tudor S.; Brinzaniuc, Klara; Petcu, Eugen B.; Miroiu, Rodica I. (2013-01-01). “Bisphosphonate-related osteonecrosis of jaw (BRONJ): diagnostic criteria and possible pathogenic mechanisms of an unexpected anti-angiogenic side effect”. Vascular Cell 5 (1): 1. doi:10.1186/2045-824X-5-1. ISSN 2045-824X. PMC 3606312. PMID 23316704.
8. Goodell, Dr. Gary G. (Fall 2012). “Endodontics: Colleagues for Excellence” (PDF). American Association of Endodontists. 211 E. Chicago Ave., Suite 1100 Chicago, IL 60611-2691: American Association of Endodontists.
9. Otto, Sven; Hafner, Sigurd; Grötz, Knut A. (2009-03-01). “The role of inferior alveolar nerve involvement in bisphosphonate-related osteonecrosis of the jaw”. Journal of Oral and Maxillofacial Surgery: Official Journal of the American Association of Oral and Maxillofacial Surgeons 67 (3): 589–592. doi:10.1016/j.joms.2008.09.028. ISSN 1531-5053. PMID 19231785.
10. Wood J, Bonjean K, Ruetz S, Bellahcene A, Devy L, Foidart JM, Castronovo V, Green JR. Novel antiangiogenic effects of the bisphosphonate compound zoledronic acid. J Pharmacol Exp Ther 2002;302:1055-61.
11. Vincenzi B, Santini D, Dicuonzo G, Battistoni F, Gavasci M, La Cesa A, Grilli C, Virzi V, Gasparro S, Rocci L, Tonini G. Zoledronic acid-related angiogenesis modifications and survival in advanced breast cancer patients. J Interferon Cytokine Res 2005;25:144-51.
12. Santini D, Vincenzi B, Dicuonzo G, Avvisati G, Massacesi C, Battistoni F, Gavasci M, Rocci L, Tirindelli MC, Altomare V, Tocchini M, Bonsignori M, Tonini G. Zoledronic acid induces significant and long-lasting modifications of circulating angiogenic factors in cancer patients. Clin Cancer Res 2003;9:2893-7
13. Chapurlat, Roland D; Arlot, Monique; Burt-Pichat, Brigitte; Chavassieux, Pascale; Roux, Jean Paul; Portero-Muzy, Nathalie; Delmas, Pierre D (2007-10-01). “Microcrack Frequency and Bone Remodeling in Postmenopausal Osteoporotic Women on Long-Term Bisphosphonates: A Bone Biopsy Study”. Journal of Bone and Mineral Research 22 (10): 1502–1509. doi:10.1359/jbmr.070609. ISSN 1523-4681.
14. Stepan, Jan J.; Burr, David B.; Pavo, Imre; Sipos, Adrien; Michalska, Dana; Li, Jiliang; Fahrleitner-Pammer, Astrid; Petto, Helmut; Westmore, Michael (2007-09-01). “Low bone mineral density is associated with bone microdamage accumulation in postmenopausal women with osteoporosis”. Bone 41 (3): 378–385. doi:10.1016/j.bone.2007.04.198. ISSN 8756-3282. PMID 17597017.
15. Woo, Sook-Bin; Hellstein, John W.; Kalmar, John R. (2006-05-16). “Narrative [corrected] review: bisphosphonates and osteonecrosis of the jaws”. Annals of Internal Medicine 144 (10): 753–761. ISSN 1539-3704. PMID 16702591.
16. Grbic JT, Landesberg R, Lin SQ; et al. (January 2008). “Incidence of osteonecrosis of the jaw in women with postmenopausal osteoporosis in the health outcomes and reduced incidence with zoledronic acid once yearly pivotal fracture trial”. J Am Dent Assoc 139 (1): 32–40. doi:10.14219/jada.archive.2008.0017. PMID 18167382.
17. Cartsos VM, Zhu S, Zavras AI (January 2008). “Bisphosphonate use and the risk of adverse jaw outcomes: a medical claims study of 714,217 people”. J Am Dent Assoc 139 (1): 23–30. doi:10.14219/jada.archive.2008.0016. PMID 18167381.
18. Zadik Y, Abu-Tair J, Yarom N, Zaharia B, Elad S. (September 2012). “The importance of a thorough medical and pharmacological history before dental implant placement.”. Aust Dent J 57 (3): 388–392. doi:10.1111/j.1834-7819.2012.01717.x. PMID 22924366.
19. Woo S, Hellstein J, Kalmar J (2006). “Narrative [corrected] review: bisphosphonates and osteonecrosis of the jaws”. Ann Intern Med 144 (10): 753–61. doi:10.7326/0003-4819-144-10-200605160-00009. PMID 16702591.
20. Wysowski D, Chang J (2005). “Alendronate and risedronate: reports of severe bone, joint, and muscle pain”. Arch Intern Med 165 (3): 346–7. doi:10.1001/archinte.165.3.346-b. PMID 15710802.
21. Black DM, Delmas PD, Eastell R; et al. (May 2007). “Once-yearly zoledronic acid for treatment of postmenopausal osteoporosis”. N. Engl. J. Med. 356 (18): 1809–22. doi:10.1056/NEJMoa067312. PMID 17476007.
21. Heckbert SR, Li G, Cummings SR, Smith NL, Psaty BM (April 2008). “Use of alendronate and risk of incident atrial fibrillation in women”. Arch. Intern. Med. 168 (8): 826–31. doi:10.1001/archinte.168.8.826. PMID 18443257.
22. Cummings SR, Schwartz AV, Black DM (May 2007). “Alendronate and atrial fibrillation”. N. Engl. J. Med. 356 (18): 1895–6. doi:10.1056/NEJMc076132. PMID 17476024.
23. “Early Communication of an Ongoing Safety Review on Bisphosphonates: Alendronate (Fosamax, Fosamax Plus D), Etidronate (Didronel), Ibandronate (Boniva), Pamidronate (Aredia), Risedronate (Actonel, Actonel W/Calcium), Tiludronate (Skelid), and Zoledronic acid (Reclast, Zometa)”. Postmarket Drug Safety Information for Patients and Providers. Food and Drug Administration (United States). 2007-10-01. Retrieved 2009-07-15.
24. “Update of Safety Review Follow-up to the October 1, 2007 Early Communication about the Ongoing Safety Review of Bisphosphonates”. Postmarket Drug Safety Information for Patients and Providers. Food and Drug Administration (United States). October 2008. Retrieved 2009-07-15.
25. Shane E (May 2010). “Evolving data about subtrochanteric fractures and bisphosphonates”. N. Engl. J. Med. 362 (19): 1825–7. doi:10.1056/NEJMe1003064. PMID 20335574.
26. Lenart BA, Lorich DG, Lane JM (March 2008). “Atypical fractures of the femoral diaphysis in postmenopausal women taking alendronate”. N. Engl. J. Med. 358 (12): 1304–6. doi:10.1056/NEJMc0707493. PMID 18354114.
27. Arkan S Sayed-Noor; Bakir K Kadum; Göran O Sjödén (31 March 2010). “Bisphosphonate-induced femoral fragility fractures: What do we know?”. Orthopedic Research and Reviews 2 (1): 27–34. doi:10.2147/ORRS7521. Retrieved 26 April 2012.
28. Frith J, Mönkkönen J, Blackburn G, Russell R, Rogers M (1997). “Clodronate and liposome-encapsulated clodronate are metabolized to a toxic ATP analog, adenosine 5′-(beta, gamma-dichloromethylene) triphosphate, by mammalian cells in vitro”. J Bone Miner Res 12 (9): 1358–67. doi:10.1359/jbmr.1997.12.9.1358. PMID 9286751.
29. van Beek E, Cohen L, Leroy I, Ebetino F, Löwik C, Papapoulos S (November 2003). “Differentiating the mechanisms of antiresorptive action of nitrogen containing bisphosphonates”. Bone 33 (5): 805–11. doi:10.1016/j.bone.2003.07.007. PMID 14623056.
30. Wiemer, AJ; Wiemer, DF; Hohl, RJ (December 2011). “Geranylgeranyl diphosphate synthase: an emerging therapeutic target.”. Clinical pharmacology and therapeutics 90 (6): 804–12. doi:10.1038/clpt.2011.215. PMID 22048229.
31. Van Beek E, Löwik C, van der Pluijm G, Papapoulos S (1999). “The role of geranylgeranylation in bone resorption and its suppression by bisphosphonates in fetal bone explants in vitro: A clue to the mechanism of action of nitrogen-containing bisphosphonates”. J Bone Miner Res 14 (5): 722–9. doi:10.1359/jbmr.1999.14.5.722. PMID 10320520.









